The Drug Administration of Vietnam, Ministry of Health has just sent a document to the Department of Health of provinces and cities regarding the counterfeit antibiotic Cefixim 200.
According to the document of the Drug Administration, this unit received an official dispatch with the Test Certificate from the Binh Duong Provincial Testing Center reporting that the product sample had the following labeling information: Cefixim 200 film-coated tablets, GĐKLH number: VD-28887-18, batch number: 15030723, NSX: 030723, HD: 030725, place of manufacture: Cuu Long Pharmaceutical Joint Stock Company.
The drug sample was taken by Binh Duong Provincial Testing Center at Bich Ngoc 1 Pharmacy (address: 395/14 Hai Ba Trung, Dong Hoa Ward, Di An City, Binh Duong Province). The drug sample did not meet the quality requirements for the qualitative index of Cefixim according to the basic standards.
Based on these results, the Drug Administration of Vietnam has sent a document to the Department of Health of provinces and centrally-run cities to strengthen the prevention of counterfeit drugs CEFIXIM 200.
Accordingly, the Drug Administration of Vietnam requires the Department of Health of provinces and cities to notify drug businesses and users of information about counterfeit drugs on the label: CEFIXIM 200 film-coated tablets, GĐKLH number: VD-28887-18; batch number: 15030723, NSX: 030723, HD: 030725, batch number: 04200623, NSX: 200623, HD: 200625 and batch number: 28201123, NSX: 201123, HD: 201125; Manufacturing facility: Cuu Long Pharmaceutical Joint Stock Company has the following characteristics and distinguishing signs from genuine drugs:

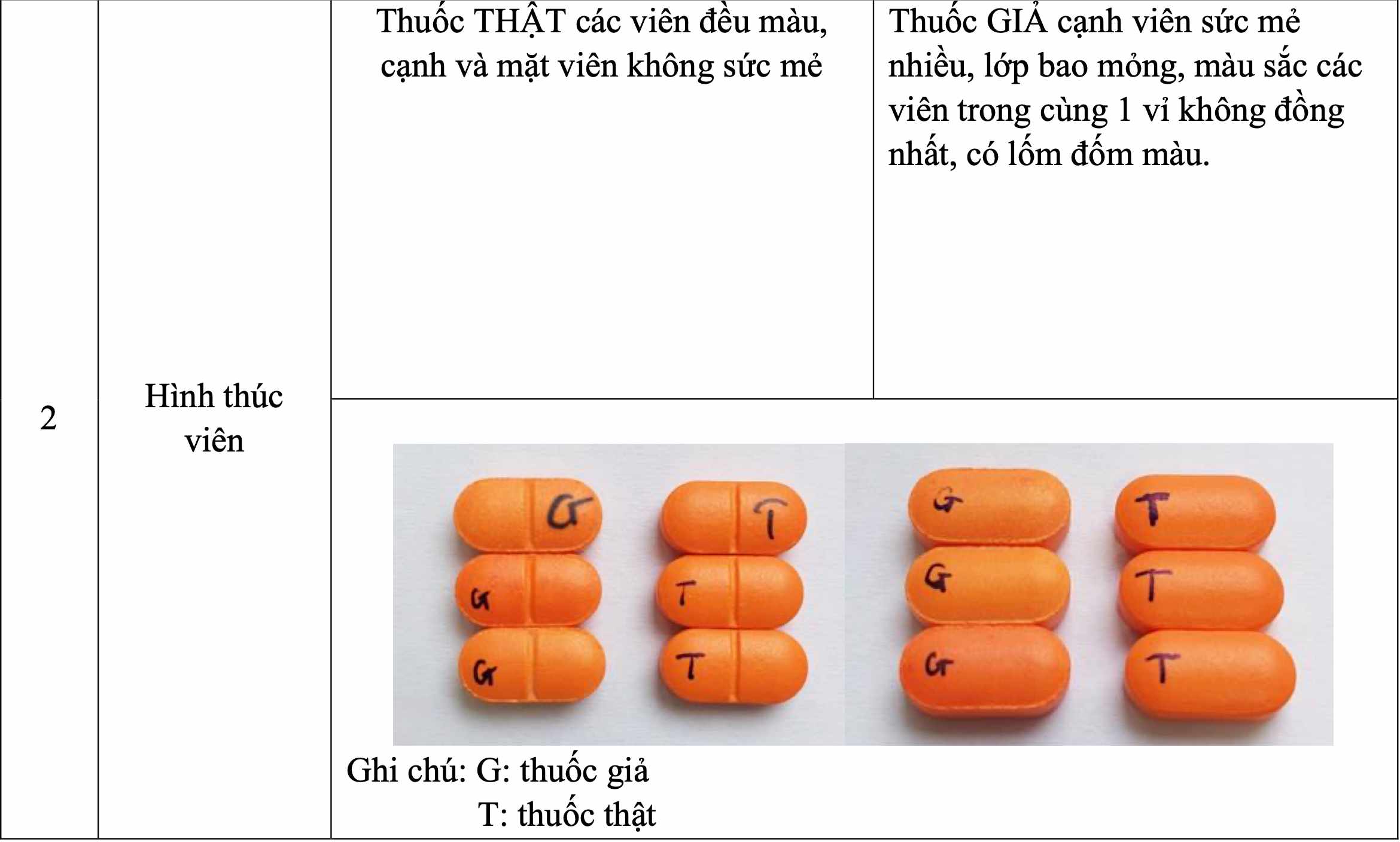
Through the above incident, the Drug Administration of Vietnam requests relevant agencies to continue to strictly implement Directive 17/CT-TTg of the Prime Minister on strengthening the fight against smuggling, trade fraud, production and trading of counterfeit and poor quality goods in the group of pharmaceuticals, cosmetics, functional foods, medicinal herbs and traditional medicine.
At the same time, strictly implement the instructions in Official Dispatch No. 7173/BYT-QLD of the Ministry of Health on strengthening management, checking drug quality, fighting against counterfeit drugs and drugs of unknown origin.
Coordinate with media agencies to inform drug trading and usage establishments and people not to trade or use fake Cefixim 200 products with the above identifying signs; only buy and sell drugs at legal pharmaceutical businesses.
Do not buy or sell drugs of unknown origin; promptly report any suspicious signs of production or trading of counterfeit drugs or drugs of unknown origin to health authorities and relevant authorities.
Coordinate with the police, market management, Steering Committee 389 and relevant authorities to conduct inspections of drug trading establishments in the area;
Investigate, verify information and trace the origin of the above fake Cefixim 200 product, handle violating organizations and individuals according to current regulations...
Cefixim 200 is a 3rd generation Cephalosporin antibiotic, used to treat bacterial infections, produced by Cuu Long Pharmaceutical. The drug is used to treat diseases such as: Otitis media, pharyngitis and tonsillitis, bronchitis, pneumonia and some cases of pyelonephritis and urinary tract infections.











