Based on the results of monitoring the quality of drugs in circulation and reviewing establishments with drugs in violation, foreign drug manufacturing establishments that are eligible to be removed from the list must take samples for quality testing of 100% of imported drug batches. The Drug Administration of Vietnam has announced the List of foreign companies with drugs in violation of quality that must take samples for quality testing of 100% of imported drug batches (pre-inspection) (Batch 39).
In which, 3 additional companies with drugs violating quality detected through post-inspection activities.
Accordingly, in this list of 46 enterprises, there are up to 43 enterprises that continue to violate from the previous announcement, adding 3 companies with drugs violating quality discovered through post-inspection activities, including the following companies: Lark Laboratories (India) Ltd (India); Kausikh Therapeutics (P) Ltd (India); Windlas Biotech Private Limited (India)
In the list of 46 violating enterprises this time, mainly Indian enterprises with 32 enterprises, the rest are some other countries such as China, Korea, USA...
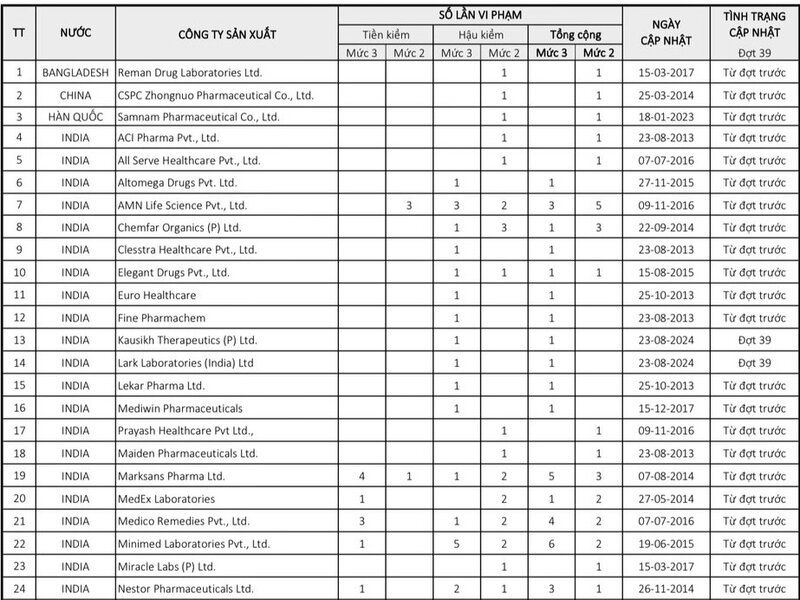
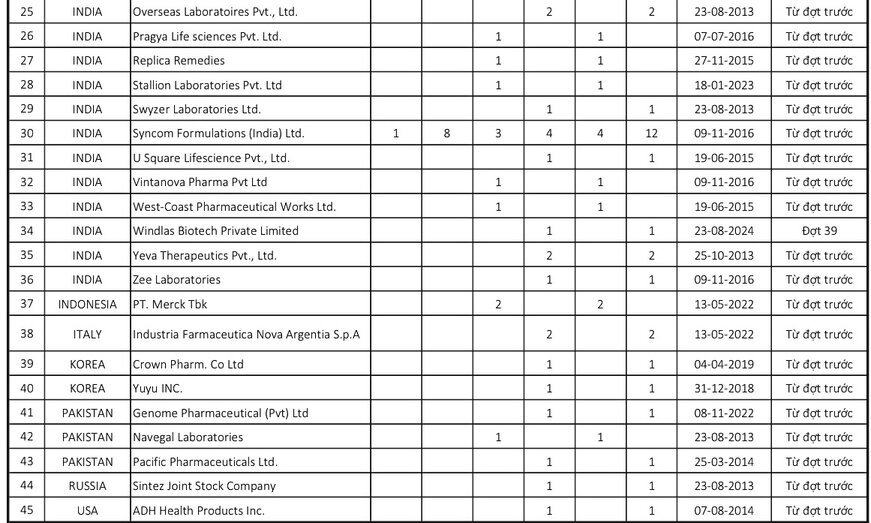
Regarding 46 enterprises with poor quality drugs, the Drug Administration of Vietnam requested the Department of Health of provinces and centrally-run cities to direct drug inspection, management and testing units under the department to inspect and supervise the implementation of regulations on quality control of imported drugs in circulation in the management area and handle organizations and individuals who violate according to current regulations.
Classification of quality violation levels:
- Level 1: Violation that causes danger that can affect the life of the drug user, cause serious injury or death.
- Level 2: Violations may affect treatment effectiveness and safety of use.
- Level 3: Violation has no or little impact on treatment effectiveness and safety of use.








