On March 24, the Drug Administration of Vietnam (Ministry of Health) issued an urgent official dispatch, warning about the appearance of the injectable drug YEZTUGO (lenacapavir) counterfeiting the Gilead Sciences Inc. brand illegally circulating on the market. This is a red alarm bell about the situation of specialized drugs being sophisticatedly counterfeited, directly threatening users' lives.
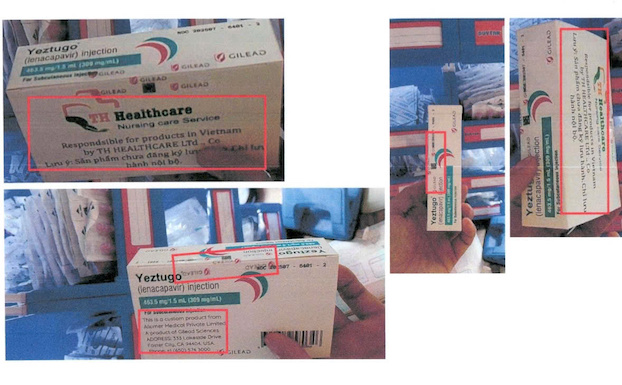
According to information from the Drug Administration of Vietnam, the incident originated from reports from Gilead Science Inc. and SNB-REACT Vietnam Co., Ltd. on the act of selling fake pre-exposure prophylaxis (PrEP). The product was discovered to be YEZTUGO (lenacapavir) injection 463. 5mg/1. 5mL (309mg/mL).
On the packaging, the subjects brazenly printed the manufacturer's name Gilead Science Inc., along with a specific address in California, USA and an international contact phone number to create absolute trust. However, through comparison on the official public service system, the Drug Administration of Vietnam affirmed that it had never issued a circulation registration certificate (GĐKLH) for this product or any production facility named YEZTUGO Injectable Drug in Vietnam.
Medical experts warn that the use of fake injections is not only economic damage but also a gamble for life. Injectable drugs of unknown origin may contain harmful impurities, not ensuring the concentration of the active ingredient lenacapavir, leading to failure in HIV prevention. More dangerously, the "underground" production process that does not ensure sterility can cause blood infection, anaphylactic shock and immediate death.
Faced with the seriousness, the Drug Administration of Vietnam requested the Ho Chi Minh City Department of Health and provinces and cities to coordinate with the police agency (C03, A03) and the National Steering Committee 389 to crack down and verify the origin of invoices and documents for buying and selling this type of medicine.
Mr. Ta Manh Hung - Deputy Director of the Drug Administration of Vietnam directed the Departments of Health of provinces and cities to immediately notify medical business establishments and people: Do not buy, sell, or use YEZTUGO (lenacapavir) injectable drugs without registration numbers.
Functional agencies will strengthen inspection, close supervision and strictly handle violating units according to current legal regulations. At the same time, testing institutes and the Ministry of Health's electronic information portal are required to publicly post information so that people can promptly prevent it.
People need to be especially alert, only buy medicine at reputable pharmaceutical establishments and look up product information on the website of the Drug Administration of Vietnam before use. If suspicious signs of fake medicine are detected, it is necessary to immediately report to functional agencies to prevent dangers.











