According to the Drug Administration, the declaration form of beauty and hair care products put on the market by SPM General Trading and Service Company Limited was revoked for the reason that there was no product information file (PIF) presented to the competent authority as prescribed.
It is known that according to the regulations of the Ministry of Health, each cosmetic product when put into circulation on the market must have a product information file (PIF - Product Information File) according to ASEAN guidelines kept at the address of the organization or individual responsible for putting the product on the market.
Cosmetic product information file includes: Administrative documents and product summary; Quality of raw materials; Quality of finished products; Safety and effectiveness.
According to regulations, the administrative documents and product summary of this Dossier must be presented immediately to the inspection and examination agency upon request; other parts, if incomplete, must be presented within 15 - 60 days from the date of inspection upon request of the competent authority.
The Cosmetic Product Declaration Receipt Number is the number issued by the competent state management agency upon receipt of the cosmetic product declaration dossier. This number is valid to certify that the cosmetic product has been declared by the organization or individual responsible for bringing the product to the market to the competent state management agency regarding the cosmetic product being circulated on the market. It is not valid to certify that the product ensures safety, effectiveness, and meets all requirements of the ASEAN Cosmetic Agreement and its Annexes.
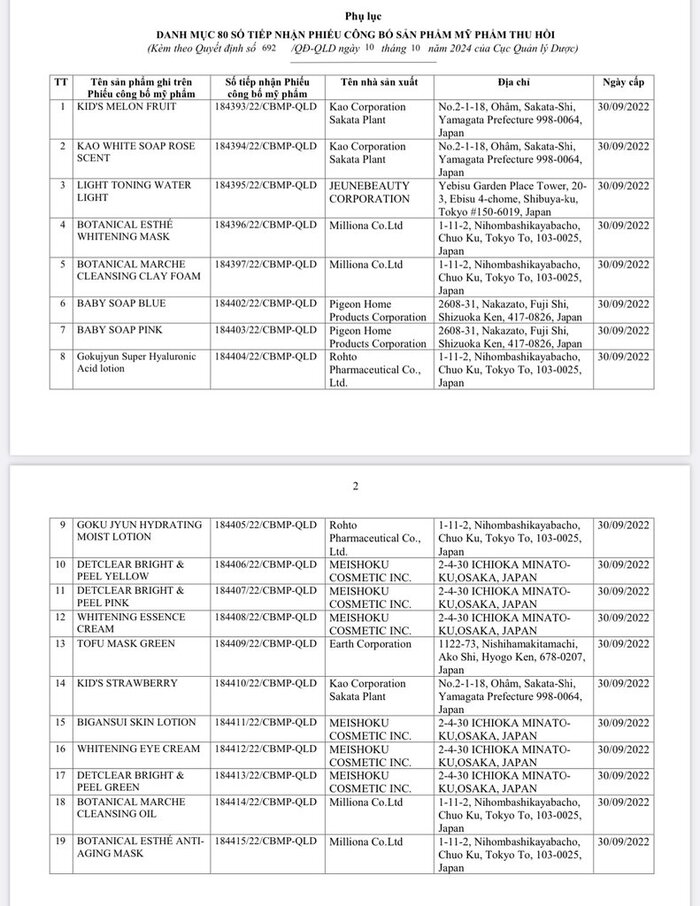
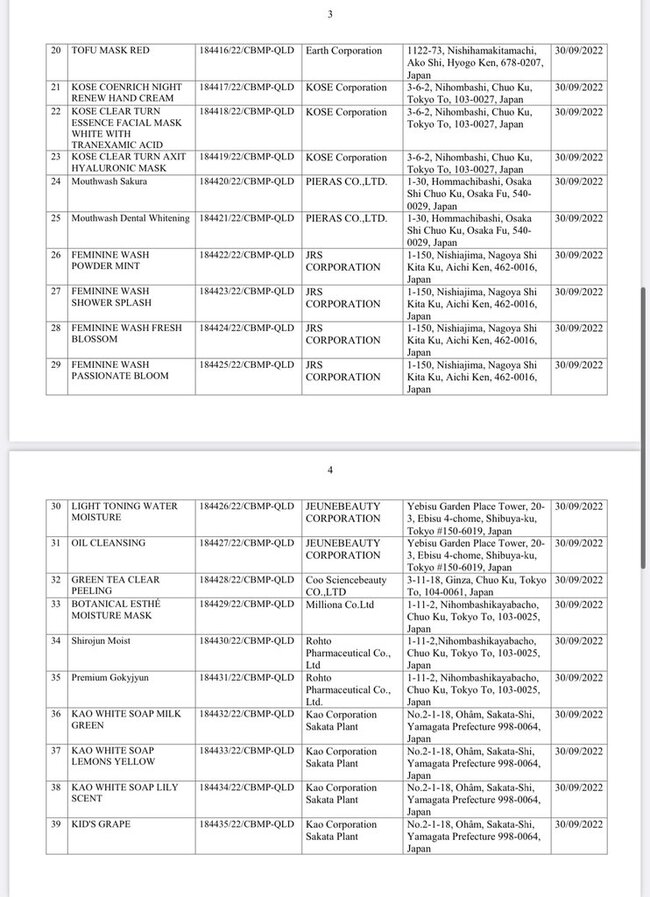
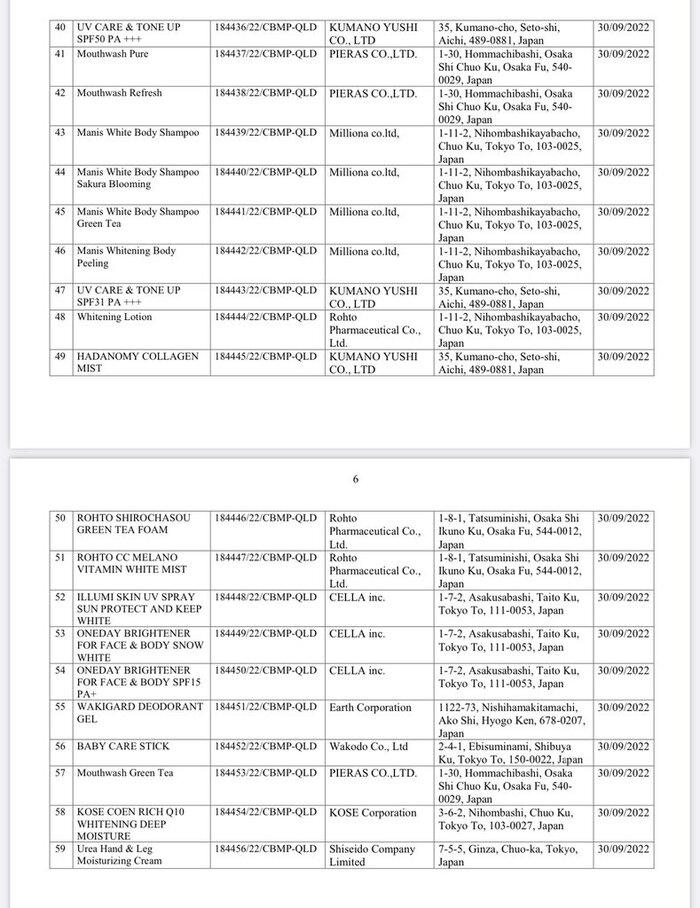
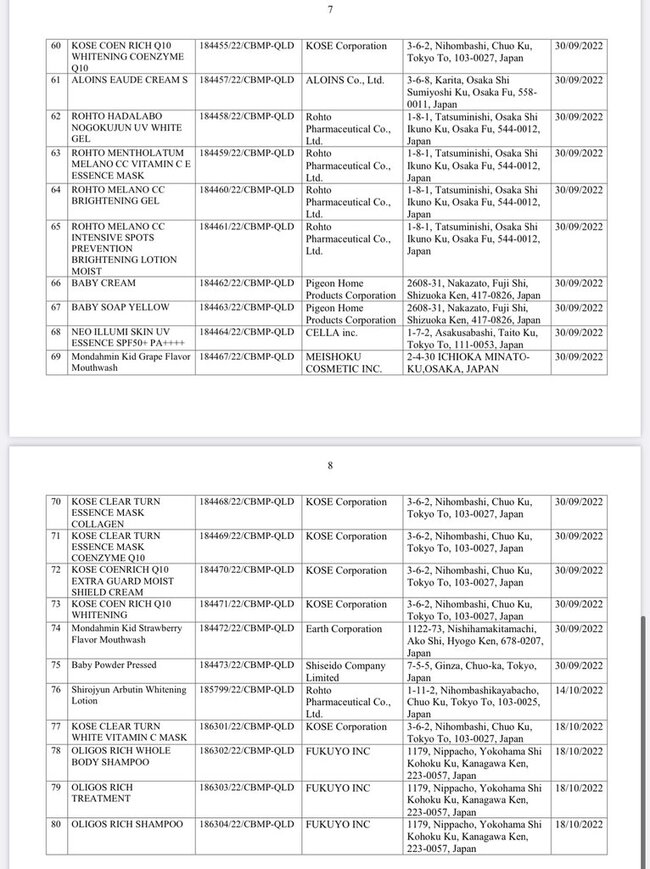
List of 80 cosmetic products with recalled Cosmetic Product Declaration Forms is as follows: