The Drug Administration of Vietnam (Ministry of Health) has just issued a Decision to revoke the number of cosmetic product declaration receipts, and at the same time suspend circulation and recall nationwide 291 cosmetic products containing two substances Cyclotetrasiloxane and Octamethylcyclotetrasiloxane.
According to the Decision, the Drug Administration of Vietnam recalled 291 batches of cosmetic product declaration receipts, suspended circulation and requested the recall of all batches of products still within their expiration dates from the list attached. These products are not allowed to continue circulating on the market from the time the decision takes effect.
The reason for the recall, as stated by the Drug Administration of Vietnam, is to comply with the regulations in the ASEAN Agreement on cosmetics. Specifically, the suspension of circulation and recall of products are carried out according to the Conclusion of the ASEAN Cosmetics Council at the 42nd session (November 20-21, 2025) and the Minutes of the 42nd ASEAN Cosmetics Science Council meeting (November 17-18, 2025) for products containing Cyclotetrasiloxane and Octamethylcyclotetrasiloxane.
The two substances mentioned above belong to the siloxane group, which is used in some personal care products. However, according to the latest update from the ASEAN regional specialized agency, products containing these two active ingredients must be suspended from circulation and recalled according to general regulations.
The Drug Administration of Vietnam requests enterprises named in the list attached to the Decision:
Send a recall notice to all distribution units, business establishments, and users of products put into circulation by the company;
Receiving returned products;
Implement the recall and destruction of all products in accordance with regulations;
Report the results of recall and destruction to the Drug Administration of Vietnam before March 26, 2026.
The Drug Administration of Vietnam also requested the Departments of Health of provinces and cities to notify cosmetic business establishments and users in the area to immediately stop trading and using the above-mentioned 291 products and return them to the supply unit;
Supervise the implementation of recovery;
Coordinate with media agencies to widely inform people not to buy, sell, or use products on the recall list;
Strictly handle violating organizations and individuals according to current regulations and report the results to the Drug Administration of Vietnam.
For localities with enterprises on the list to be recalled, the Department of Health is responsible for supervising the recall and destruction of products and reporting the inspection and supervision results to the Drug Administration of Vietnam before April 1, 2026.
The widespread recall of 291 cosmetic products this time is considered a strong move to ensure compliance with the general regulations of the ASEAN region, while protecting the rights and safety of consumers.
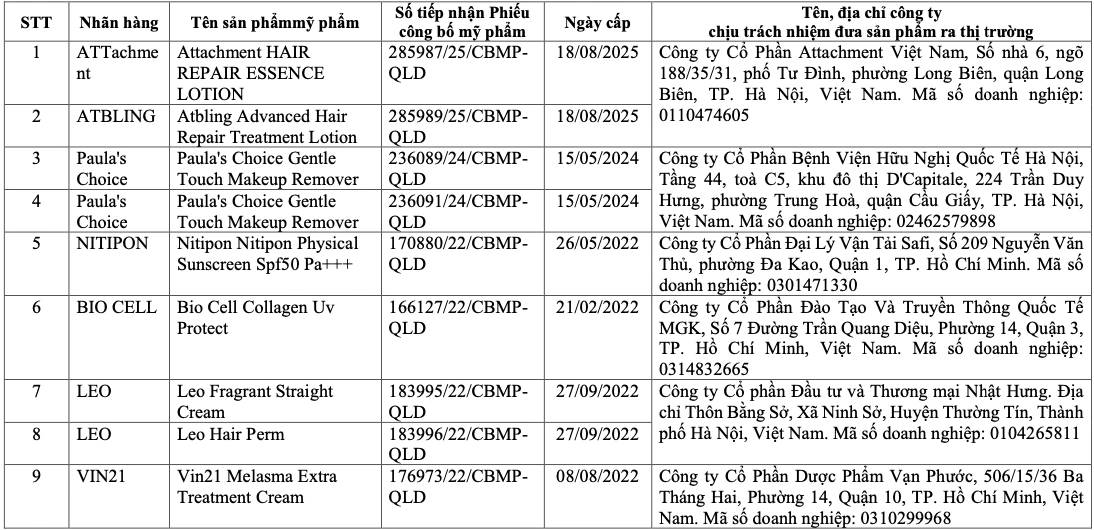
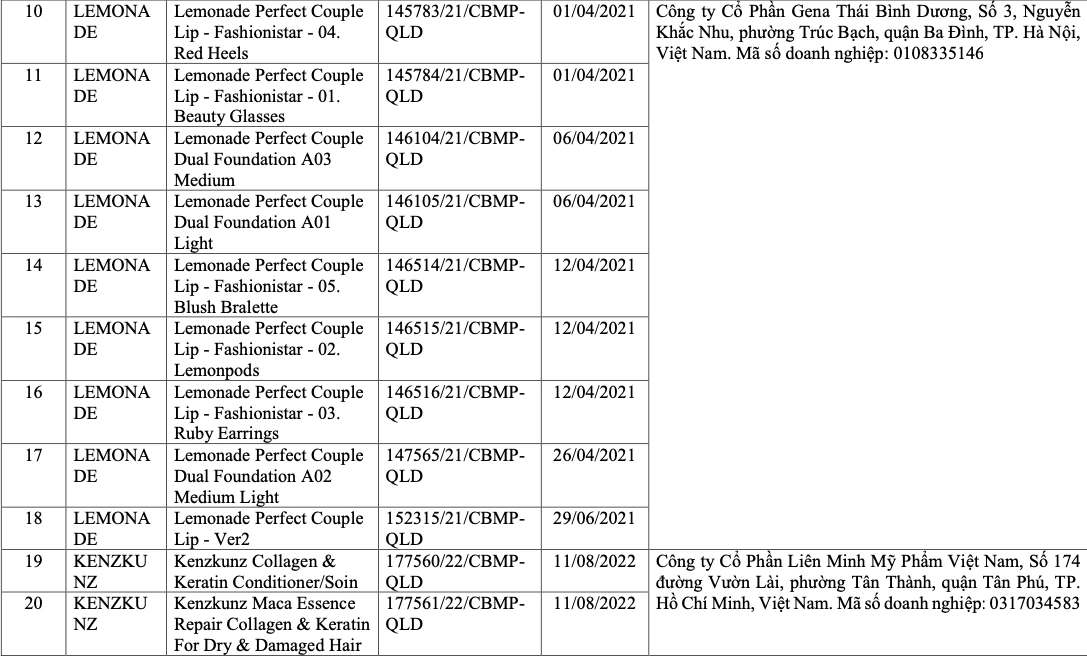
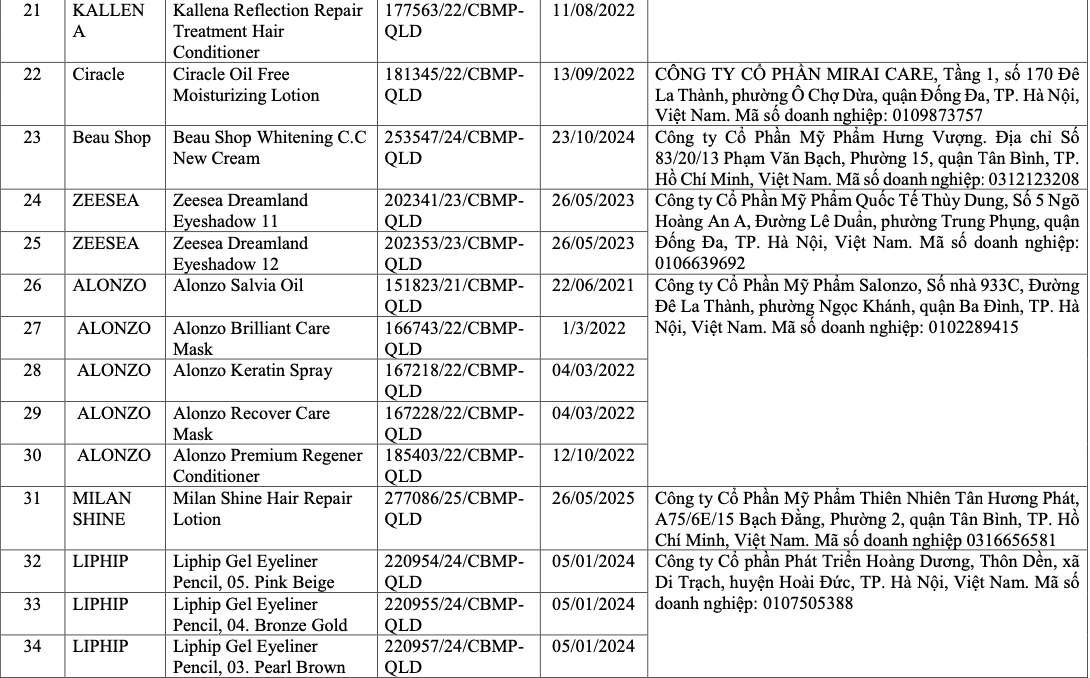
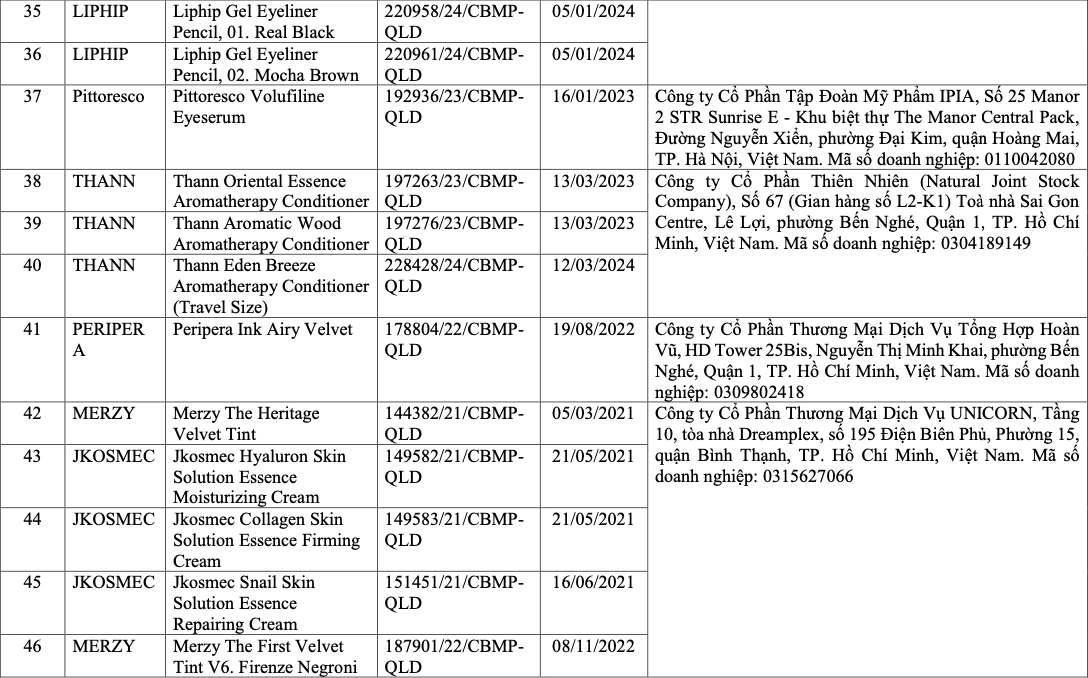
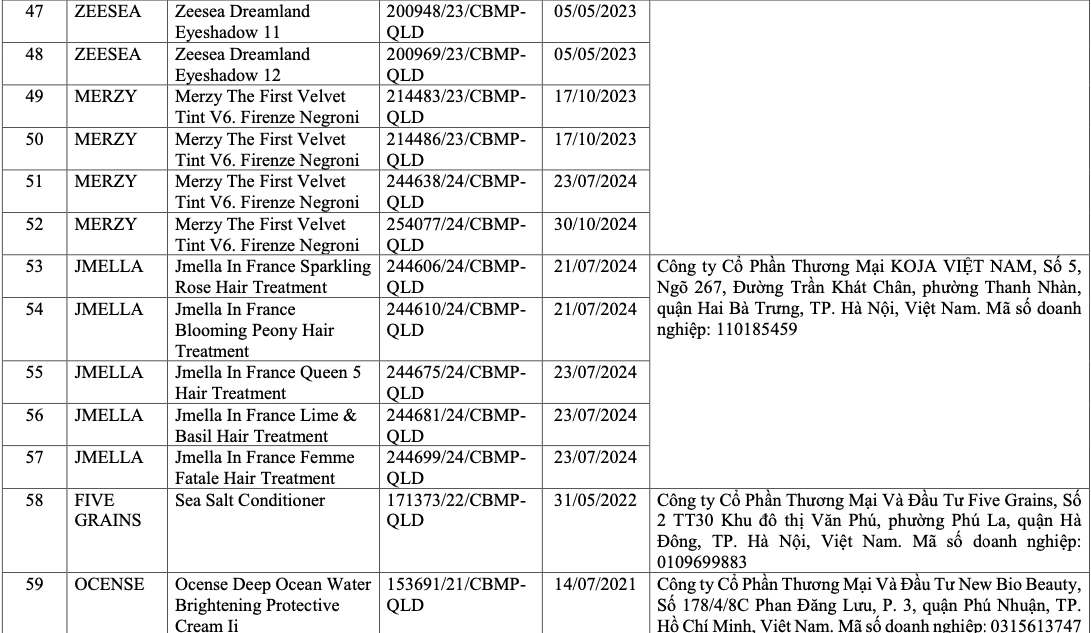
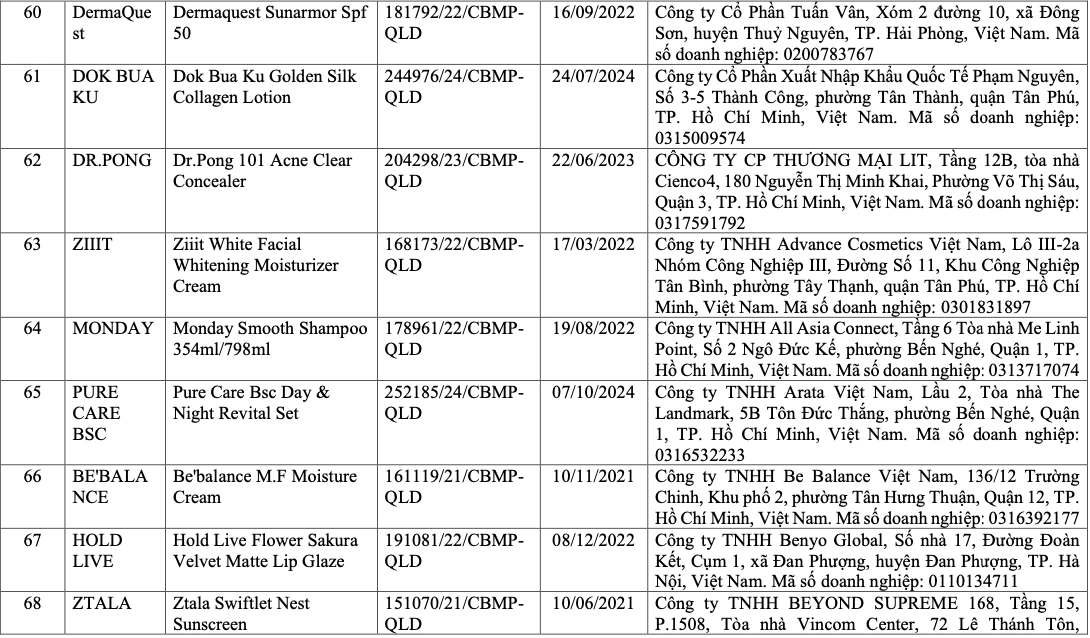
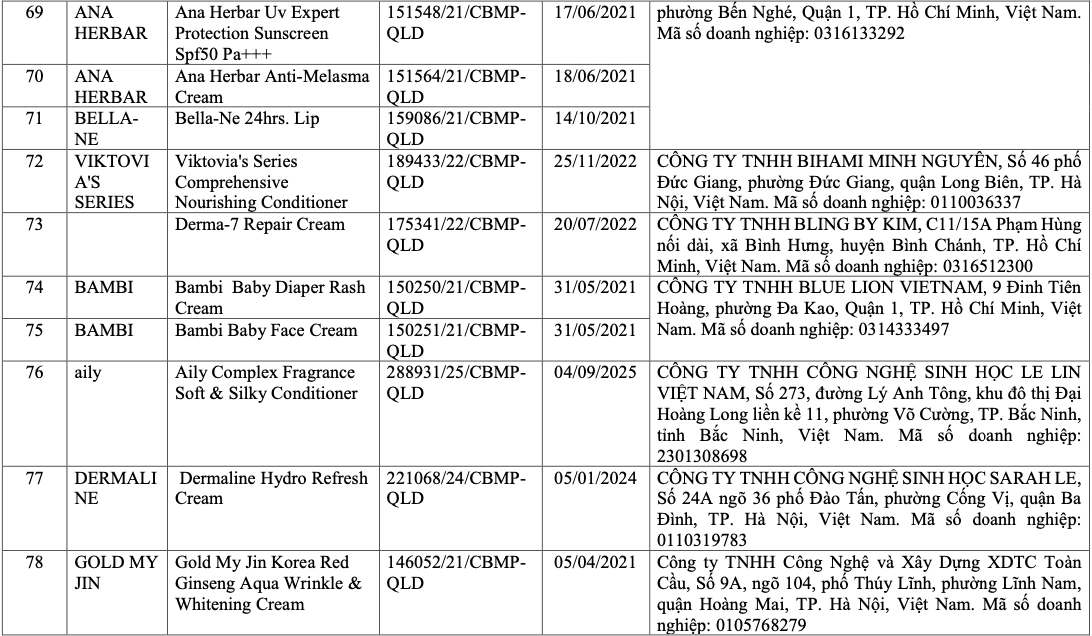
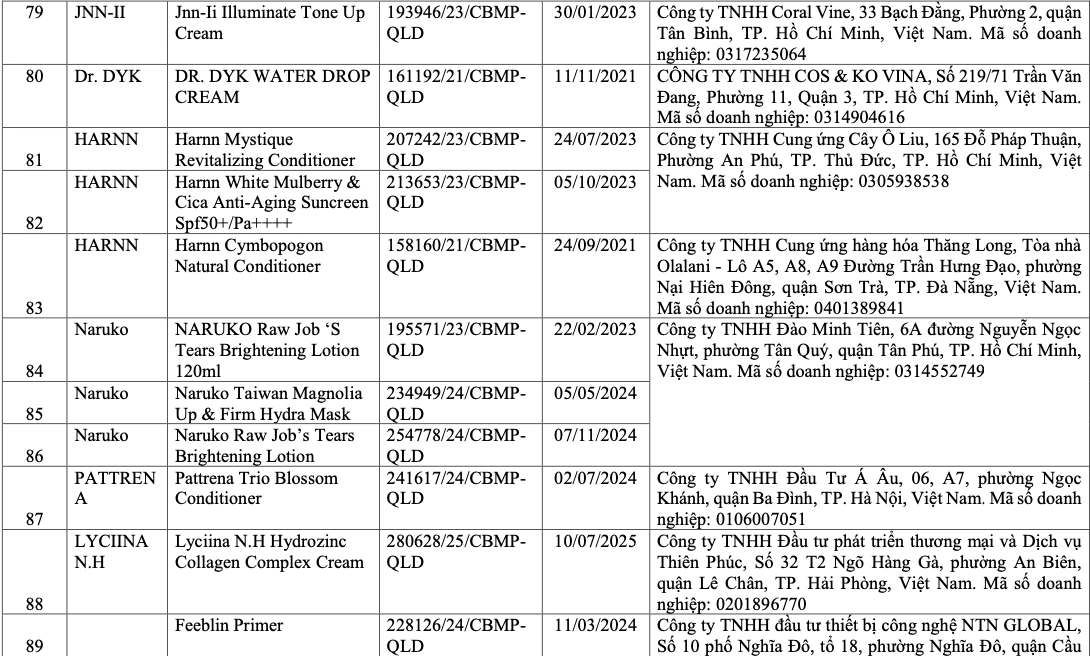
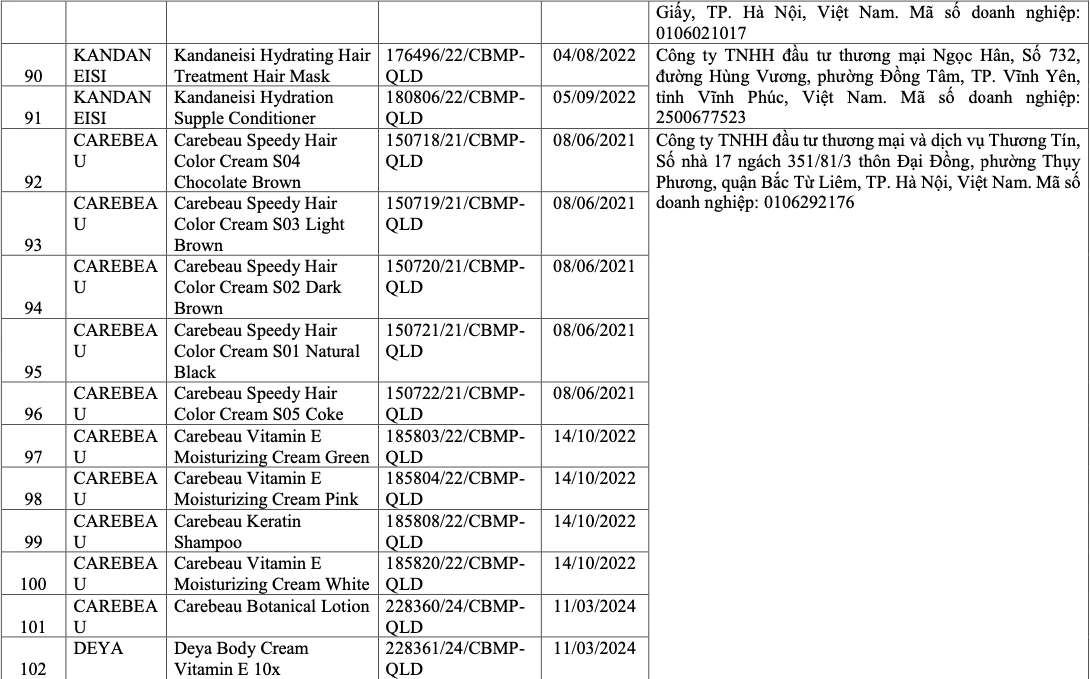
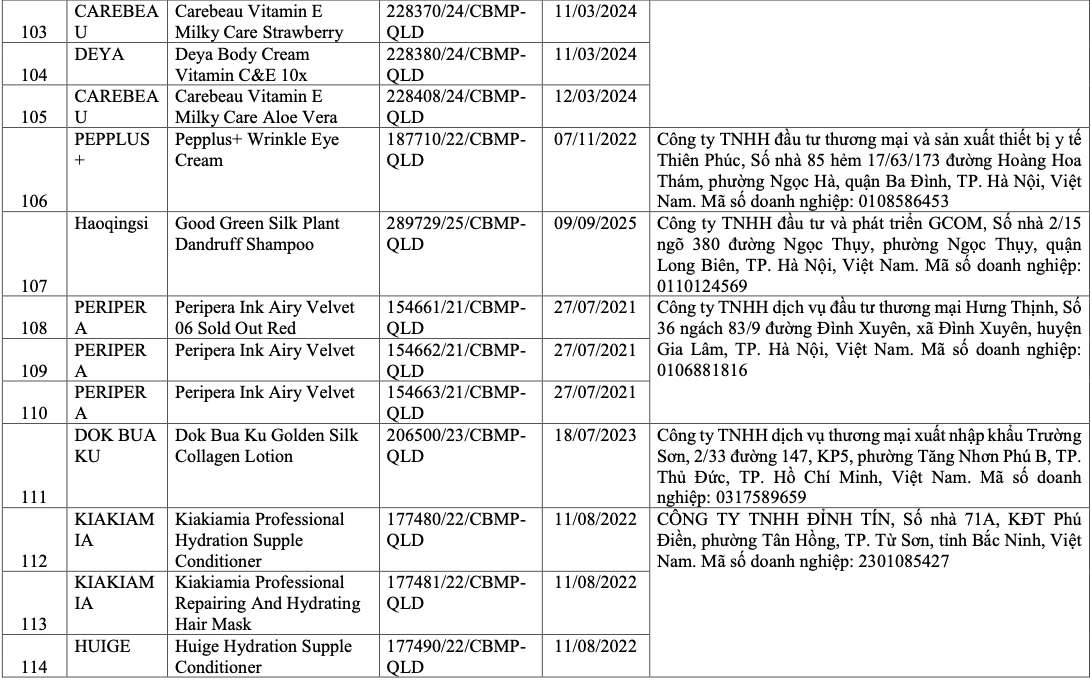
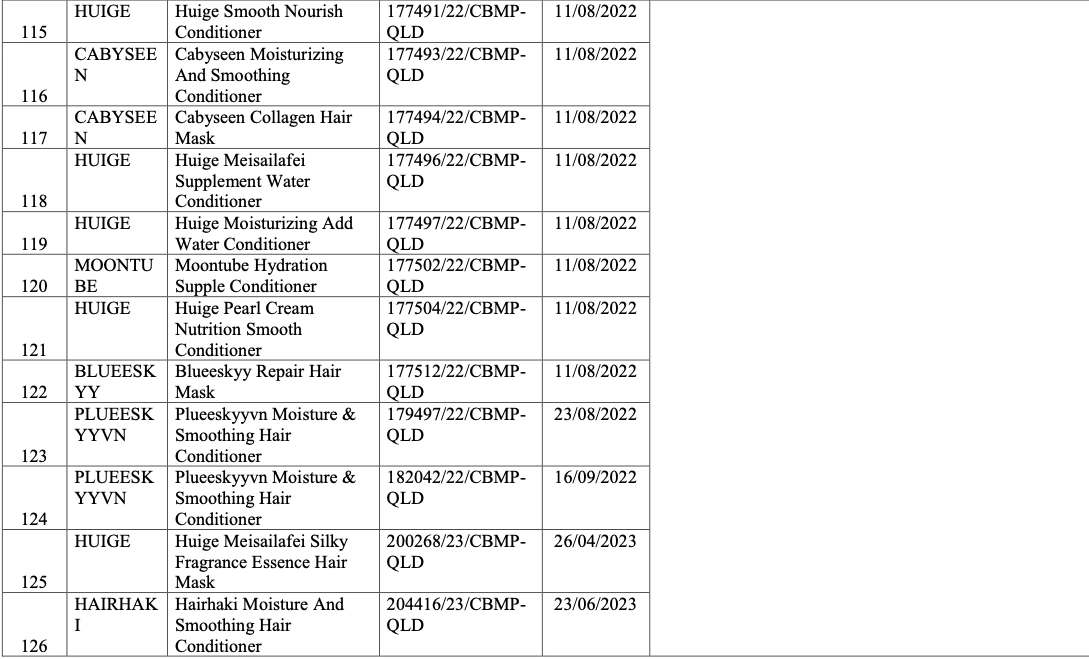
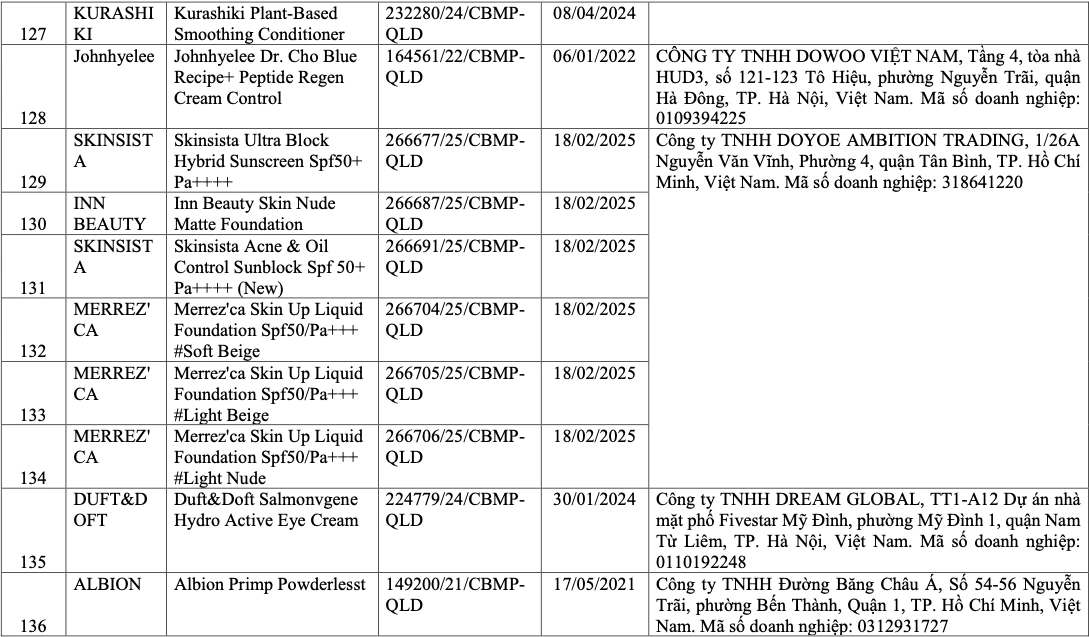
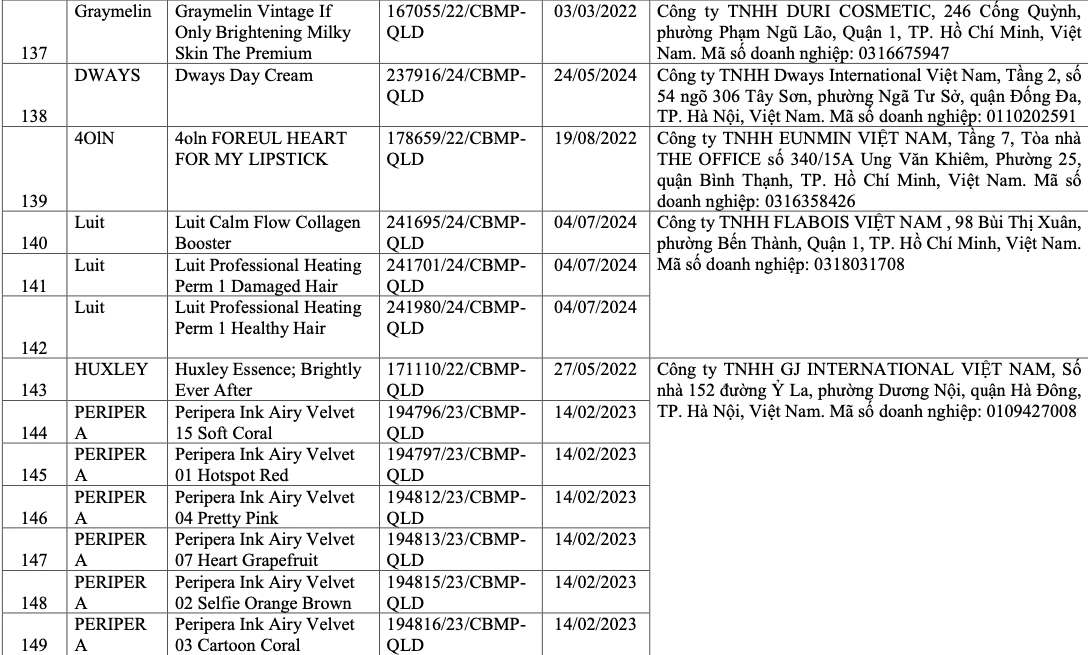
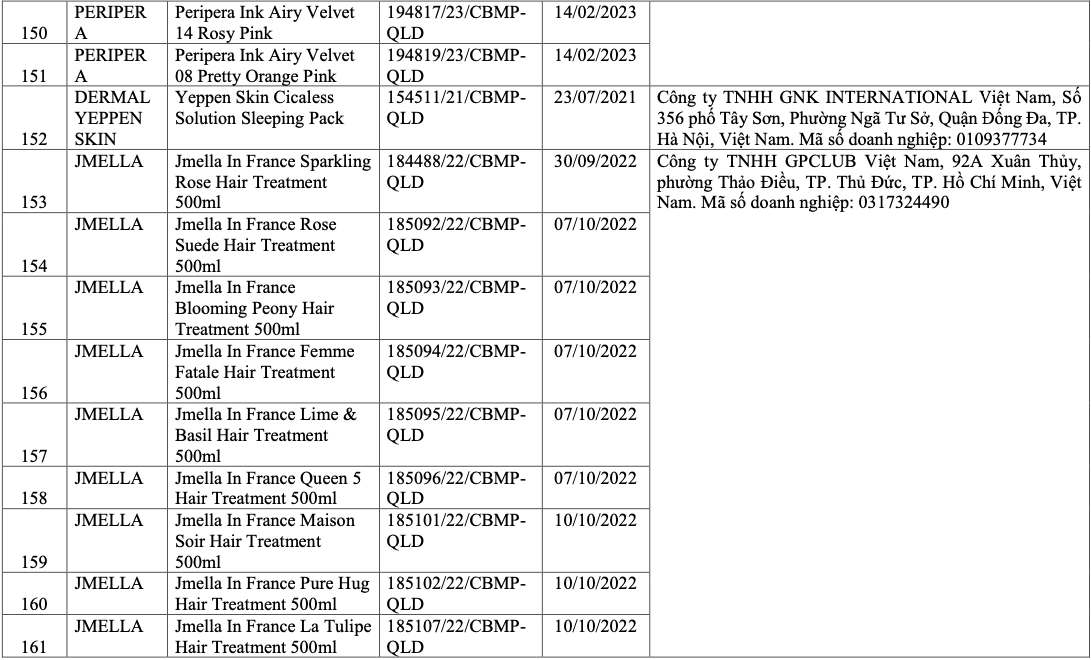
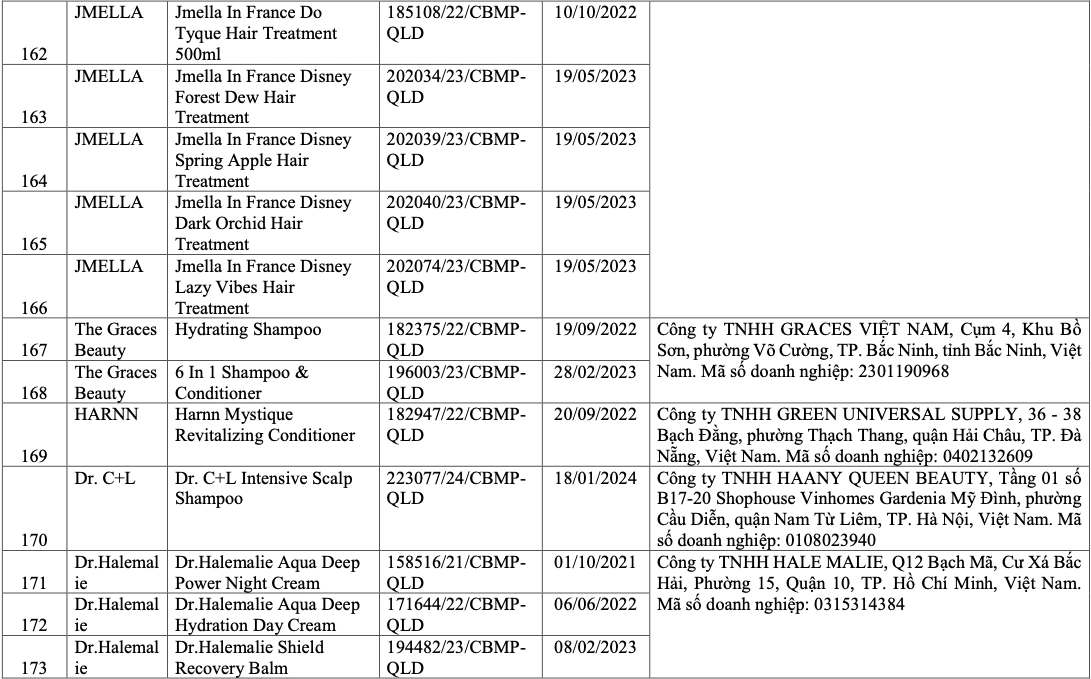
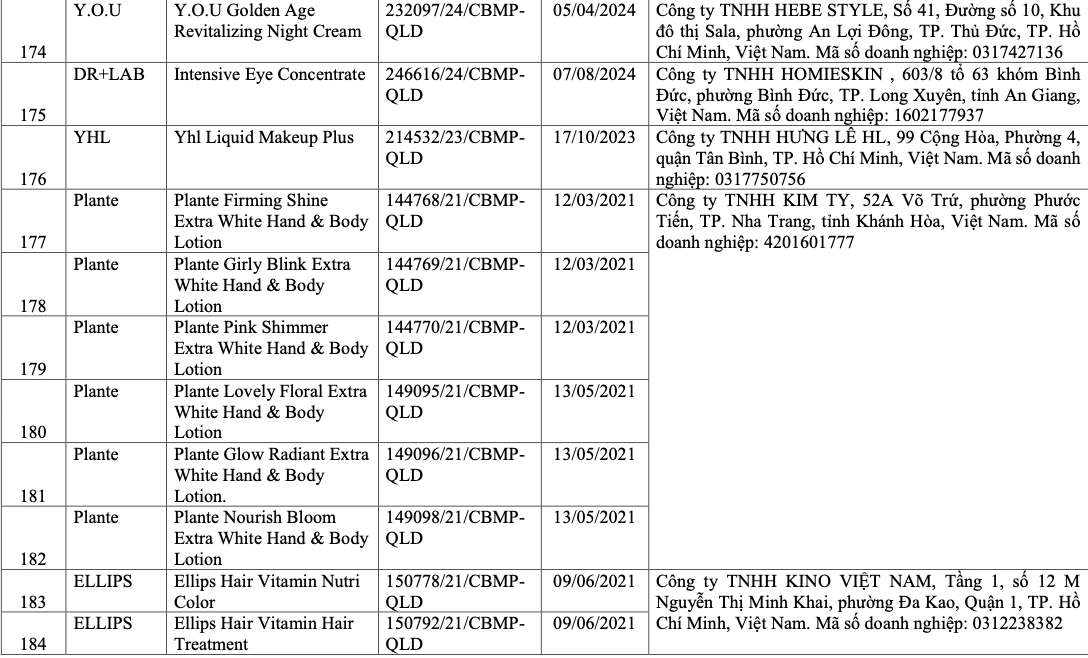
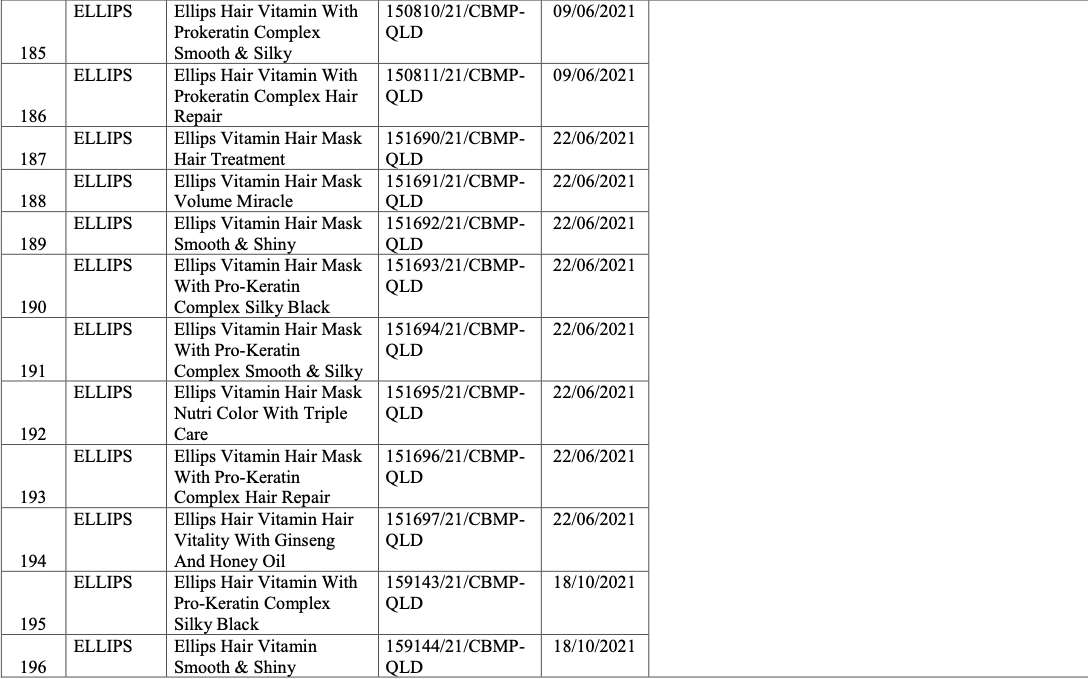
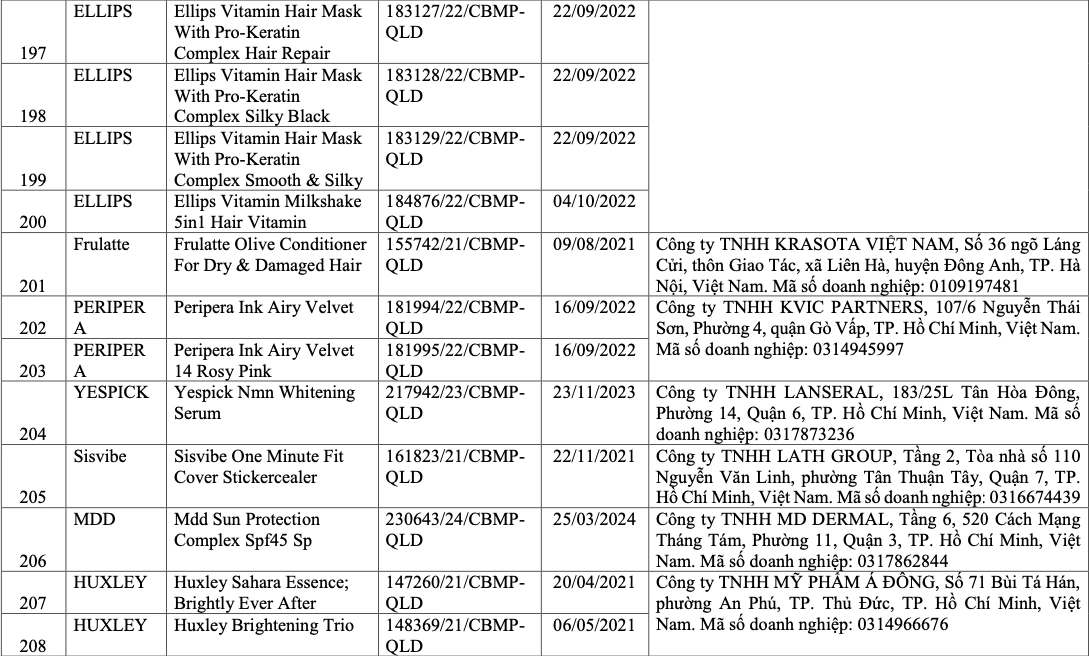
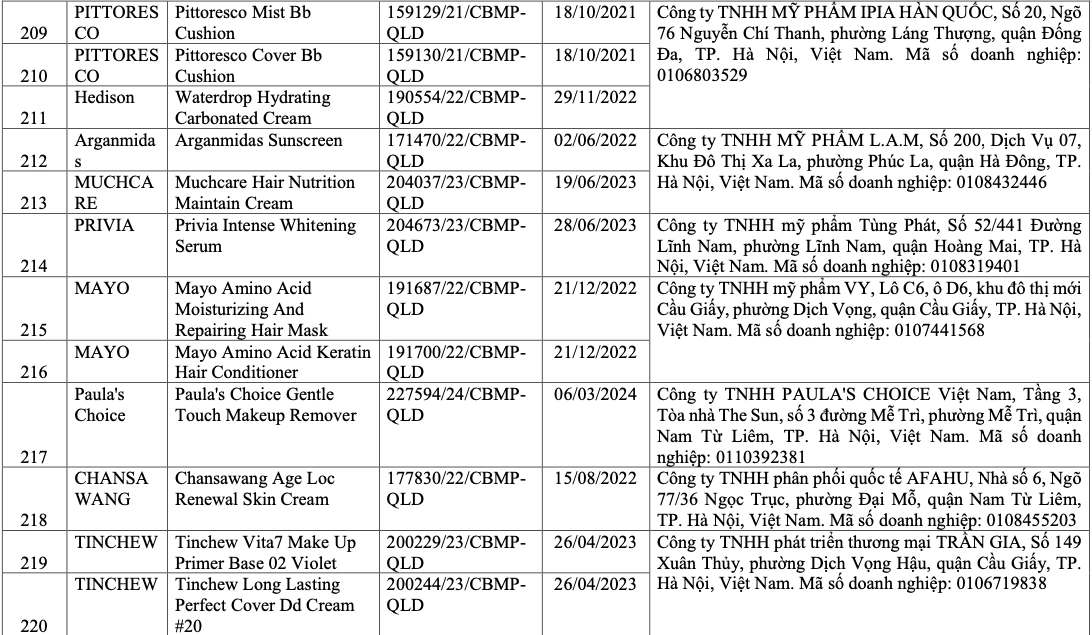
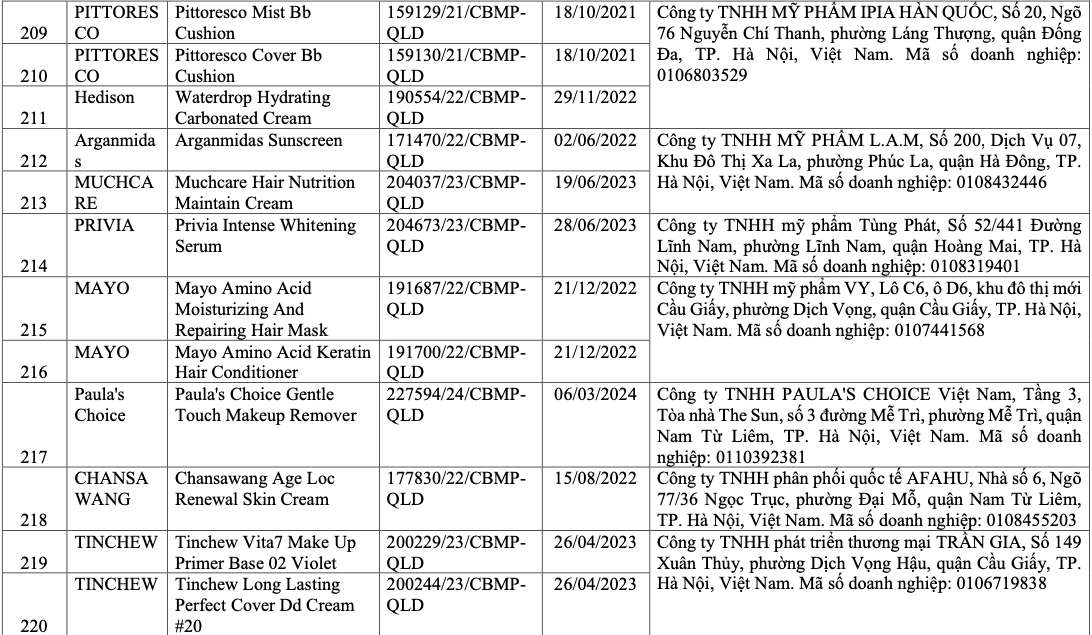
List of recalled cosmetics: